
This study identifies the potential of non-vdW Cr 2S 3 toward new and robust 2D-based spintronic applications. The computed r M magnitude is tenfold higher than the bulk Cr 2S 3. These prototype Al/Cr 2S 3/Al structures have demonstrated a low power (≈2 µW for 8 µm channel at 10 ♚ current) operation with a negative field-dependent resistance coefficient ( r M) of ≈−5 × 10 −4 Oe −1. Chromium sulfides are usually nonstoichiometric compounds. What is the formula for chromium (III) fluoride The formula for chromium (III) fluoride is CrF3. The magnetic field-induced spin polaron formations are quite resilient and enhance the field-dependent carrier conduction, making it greatly useful for negative magnetoresistance operation. The sorption of chromium(III) and chromium(VI) on lead sulfide has been investigated in dependence on pH, time of sorption and the concentrations of sorbat. Chromium(III) sulfide is the inorganic compound with the formula CrS. The prototype lateral spin-channel Al/Cr 2S 3/Al devices exhibit a negative magnetoresistance of ≈25% and ≈15% at 100 K (below Néel temperature) for few-layer (≈8 nm) and thin-film (≈50 nm) device structures, respectively, owing to itinerant ferrimagnetism of Cr 2S 3. Find molecular formula and molecular weight of chromium(iii) sulfide or Find chemical formula or molecular formula of different substances, calculate its. 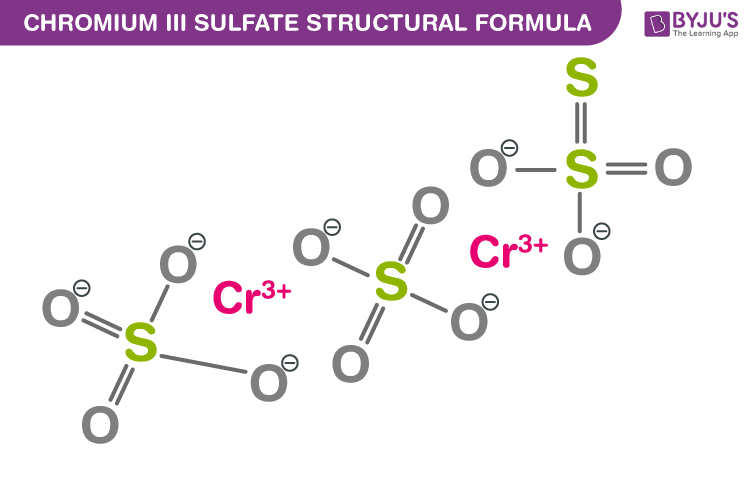
Importantly, the anisotropic magnetic saturation and resistivity of 10.23 mΩ cm reveal a narrow bandgap ferrimagnetism in Cr 2S 3 (∆ E Activation ≈ 22 meV and E g ≈ 40 meV). The sorption of chromium(III) and ehromium(VI) on lead sulfide has been investigated in dependence on pH, time of sorption and the concentrations of sorbate. This report demonstrates a facile, bottom-up, chemical vapor deposition approach for non-van der Waals (non-vdWs), 2D chromium (III) sulfide (Cr 2S 3) with c-axis orientation, out-of-plane magnetic anisotropy (easy axis), and ferrimagnetic ordering. Retrieved 1 November 2021.Magnetic 2D materials have emerged as a great interest in spintronics due to atomic thickness scaling, low energy switching, and ease in the manipulation of spins. (II)+sulfide+CrS&pg=PA3082&printsec=frontcover. Download Citation Chromium sulfide nanoparticles as an ion-selective membrane electrode for chromium(III) Nanoparticles of chromium sulfide were. ↑ Riedel, Erwin Janiak, Christoph (2011) (in German).(II)+sulfide&pg=SA6-PA111&printsec=frontcover. (II)+sulfide&pg=PA670&printsec=frontcover. (II)+sulfide+CrS&pg=PA1377&printsec=frontcover. It means that the chromium atom loses three electrons from its valence shell and thus exhibits +. Chromium sulfides are usually nonstoichiometric compounds. (II)+sulfide+CrS&pg=PA1074&printsec=frontcover. In Chromium(III) sulfide, Chromium atoms have +3 charge on it. Chromium sulfide powder is the inorganic compound with the formula Cr2S3. Bretherick's Handbook of Reactive Chemical Hazards. It may also be formed by reacting chromium(III) chloride with H 2S, reducing chromium(III) sulfide with hydrogen, or by double replacement reaction of lithium sulfide with chromium(II) chloride. Chromium(II) sulfide may be formed by reaction of chromium metal with sulfur or hydrogen sulfide at high temperature.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
